
Cancer immunotherapy has transformed the treatment landscape of oncology by shifting the therapeutic paradigm from direct tumor targeting to modulation of the host immune system. As summarized in the review by Arifa Aman et al., “The Broad Spectrum of Cancer and Immunotherapy: Achievements and Limitations,” modern immunotherapy can be broadly divided into three major strategies: immune checkpoint blockade, adoptive cell therapy, and cancer vaccines.
While these approaches have demonstrated unprecedented clinical success, particularly in melanoma, lung cancer, and hematologic malignancies, their effectiveness remains heterogeneous, and significant biological and clinical challenges persist.
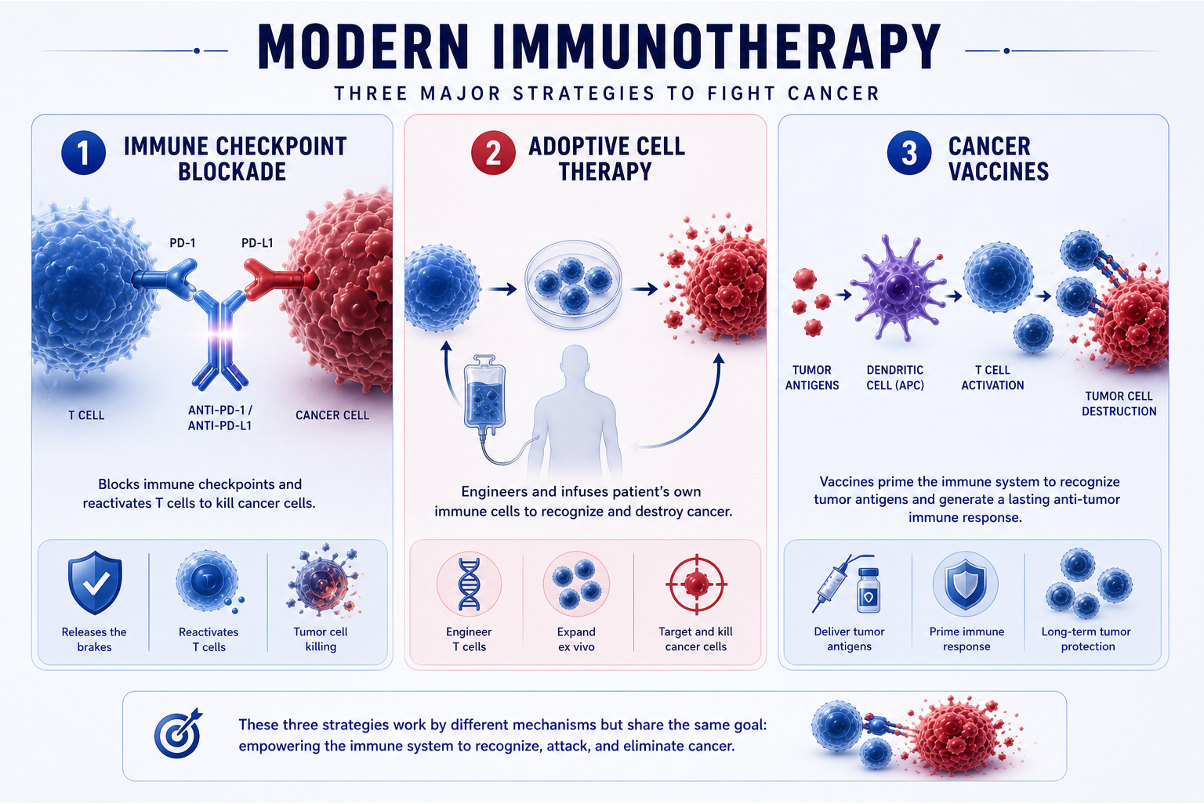
Immune checkpoint inhibitors (ICIs) represent the most widely used and clinically validated form of immunotherapy. By blocking inhibitory pathways such as PD-1/PD-L1 and CTLA-4, these agents restore T-cell–mediated antitumor immunity.
Clinically, drugs such as:
•Pembrolizumab,
•Nivolumab,
•Ipilimumab,
have demonstrated durable responses across multiple tumor types.
However, the limitations of checkpoint blockade are clearly illustrated by clinical trial data. In the CheckMate 650 trial, which evaluated nivolumab plus ipilimumab in metastatic castration-resistant prostate cancer, the objective response rate was 25% in chemotherapy-naïve patients and 10% in previously treated patients, highlighting modest efficacy in this tumor type. Importantly, treatment-related grade ≥3 toxicities occurred in 42–53% of patients, demonstrating that increased immune activation is closely linked to increased toxicity.
This duality reflects a central principle of immunotherapy:
The same immune activation responsible for tumor control can also drive toxicity and limit tolerability.
Although immune checkpoint inhibitors have transformed cancer treatment, only a subset of patients derive meaningful benefit. A major challenge in clinical practice is the development of resistance, which can occur in two main forms.
Primary resistance refers to the absence of an initial response to therapy.
Acquired resistance develops after an initial response, when the disease later progresses despite ongoing treatment.
These resistance patterns reflect underlying biological mechanisms that limit effective immune activation.
Several key factors contribute to resistance:
•Impaired antigen presentation
Tumor cells may lose or downregulate molecules required for immune recognition, such as MHC complexes or components like B2M, preventing T cells from identifying cancer cells.
•Tumor-intrinsic signaling pathways
Oncogenic pathways, including MAPK activation or mutations such as KEAP1, can promote immune evasion and reduce sensitivity to immunotherapy.
•Immunosuppressive tumor microenvironment (TME)
The presence of regulatory T cells, myeloid-derived suppressor cells, and inhibitory cytokines creates a local environment that suppresses effective immune responses.
•Low tumor mutational burden (TMB)
Tumors with fewer mutations generate fewer neoantigens, making them less visible to the immune system and less responsive to checkpoint blockade.
These mechanisms are particularly evident in tumors such as pancreatic cancer and microsatellite-stable colorectal cancer, which remain largely non-responsive to single-agent checkpoint inhibitors.
Adoptive cell therapy (ACT) represents one of the most personalized strategies in oncology, based on the ex vivo isolation, modification, and expansion of immune cells, followed by their reinfusion to enhance antitumor activity. While this approach offers a high level of precision, it is also associated with significant biological and logistical complexity.
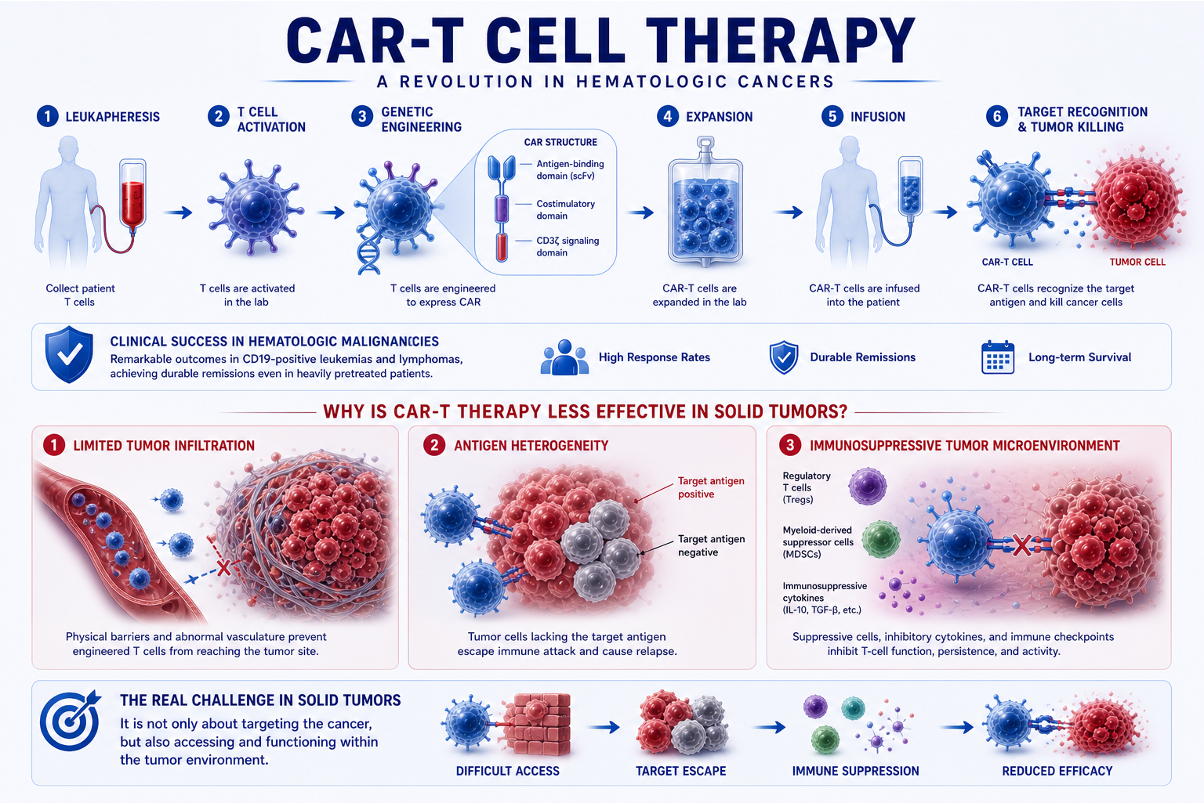
CAR-T Cell Therapy
Chimeric antigen receptor T-cell (CAR-T) therapy has achieved remarkable clinical success in hematologic malignancies, particularly in CD19-positive leukemias and lymphomas, where durable remissions have been observed even in heavily pretreated patients.
However, extending this success to solid tumors has proven challenging. Several key barriers limit efficacy:
•Limited tumor infiltration, preventing engineered T cells from reaching the tumor site
•Antigen heterogeneity, allowing tumor cells lacking the target antigen to escape immune attack
•Immunosuppressive tumor microenvironment (TME), which inhibits T-cell function and persistence
These factors highlight that, in solid tumors, the issue is not only targeting the cancer, but also accessing and functioning within the tumor environment.
A significant recent advance in adoptive cell therapy is the development of tumor-infiltrating lymphocyte (TIL) therapy, which utilizes naturally occurring tumor-reactive T cells isolated directly from the tumor.
One of the most important examples is lifileucel, evaluated in the C-144-01 phase II trial in patients with advanced melanoma previously treated with checkpoint inhibitors.
Key results from this study include:
•Objective response rate: 31.5%
•Complete responses: approximately 4%
•Durable responses, with long-term persistence in a subset of patients
These findings are clinically meaningful because they demonstrate that cell-based therapies can induce responses even in tumors resistant to checkpoint inhibition.
Adoptive cell therapy illustrates both the strength and limitations of precision immunotherapy. On one hand, it enables highly targeted and potentially durable immune responses. On the other, its widespread application is constrained by:
•Complex and time-intensive manufacturing
•Need for specialized infrastructure
•Treatment-related toxicities
•Variability in patient-specific cell products
Overall, ACT represents a powerful but still evolving approach, with ongoing efforts focused on improving scalability, safety, and efficacy in solid tumors.
Cancer vaccines were historically limited by modest clinical efficacy, but recent advances in mRNA platforms, neoantigen identification, and combination strategies have significantly renewed interest in this field. Current approaches are no longer based on single-antigen targeting alone, but aim to generate broad, tumor-specific T-cell responses, often in combination with immune checkpoint inhibitors.
A major breakthrough in this area is the KEYNOTE-942 trial, led by Jeffrey Weber, which evaluated the personalized mRNA vaccine mRNA-4157/V940 in combination with Pembrolizumab in patients with resected high-risk melanoma.
Key findings include:
•Approximately 49% reduction in the risk of recurrence or death
•Recurrence-free survival of 74.8% with the combination vs 55.6% with pembrolizumab alone
These results provide strong clinical evidence that personalized neoantigen vaccines can enhance the efficacy of checkpoint blockade, particularly in the adjuvant setting where tumor burden is minimal.
Another important example comes from the phase 1 study by Vinod Balachandran (Rojas et al.), which evaluated the individualized mRNA vaccine autogene cevumeran in resected pancreatic ductal adenocarcinoma.
Key observations:
•50% of patients developed neoantigen-specific T-cell responses
•Patients with immune responses showed significantly improved relapse-free survival
This is particularly notable because pancreatic cancer is typically considered immunologically resistant, yet vaccine-induced T-cell activation was clearly associated with improved clinical outcomes.
An alternative strategy is to stimulate immunity directly within the tumor, effectively turning the tumor into its own vaccine. This concept has been clinically validated with Talimogene laherparepvec (T-VEC), evaluated in the OPTiM phase 3 trial by Robert Andtbacka.
Key results:
•Durable response rate: 16.3% vs 2.1% (T-VEC vs GM-CSF)
•Overall survival: 23.3 vs 18.9 months
These findings demonstrate that local intratumoral therapy can generate systemic antitumor immune responses, including regression of distant metastases.
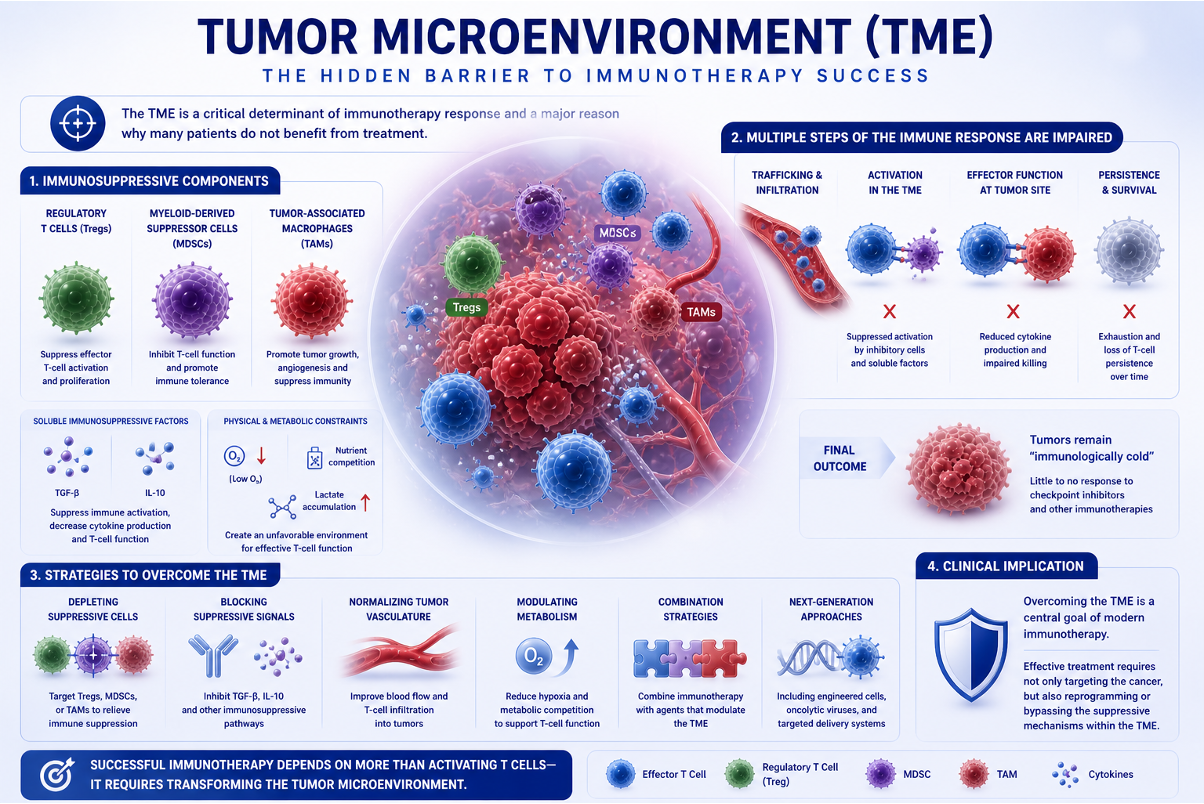
The tumor microenvironment (TME) is a critical determinant of immunotherapy response and one of the main reasons why many patients do not benefit from treatment. Even when tumor-specific T cells are generated, their activity can be significantly limited by the local environment surrounding the tumor.
Several immunosuppressive components contribute to this effect. These include regulatory T cells (Tregs), myeloid-derived suppressor cells (MDSCs), and tumor-associated macrophages (TAMs), all of which actively inhibit antitumor immunity. In addition, soluble factors such as TGF-β and IL-10 further suppress immune activation, while hypoxia and metabolic competition within the tumor create conditions that are unfavorable for effective T-cell function.
As a result, multiple steps of the immune response are impaired. T cells may fail to adequately infiltrate the tumor, become functionally suppressed upon arrival, or lose their persistence over time. These combined effects explain why many tumors remain “immunologically cold”, showing little response to checkpoint inhibitors and other immunotherapies.
From a clinical perspective, overcoming the tumor microenvironment has become a central goal of modern immunotherapy, with ongoing strategies focused on reprogramming or bypassing these suppressive mechanisms.
While immunotherapy can produce durable antitumor responses, it is also associated with immune-related adverse events (irAEs), reflecting unintended activation of the immune system against normal tissues.
These toxicities can affect multiple organ systems. Common manifestations include:
• Endocrine disorders, such as thyroid dysfunction or hypophysitis
• Gastrointestinal toxicity, particularly immune-mediated colitis
• Hepatic toxicity, including hepatitis
• Pulmonary toxicity, such as pneumonitis
Management typically involves corticosteroids or other immunosuppressive agents, and in severe cases, treatment interruption or discontinuation may be required.
Importantly, toxicity should not be viewed solely as a limitation. In many cases, the development of irAEs reflects a biologically active immune response, indicating that the immune system has been effectively engaged. This creates a complex clinical balance, where the goal is to maximize antitumor immunity while minimizing immune-mediated damage.
About IMMONC
Immune Oncology Research Institute (IMMONC) is dedicated to advancing research aimed at preventing, treating, and ultimately curing cancer while making these innovations accessible to those who need them. If you're interested in joining our team, please feel free to contact us at [email protected] or at +374-41 310-048.